Golden Oyster Mushroom (Pleurotus citrinopileatus)

French common name: Pleurote jaune
Family: Pleurotaceae
Order: Agaricales
Did you know? Oyster mushrooms (genus Pleurotus) are carnivorous fungi that can secrete toxins to paralyze and then consume nematodes (microscopic roundworms) to supplement their nitrogen intake (Lee et al. 2023).
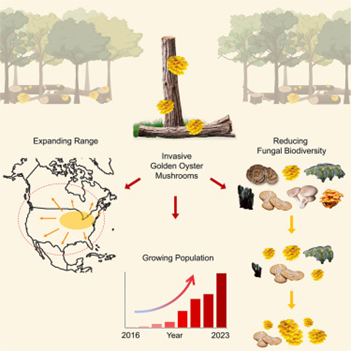
The Golden Oyster Mushroom (GOM) is a saprotrophic, wood-decaying, and aggressively spreading fungus that grows in dense clumps primarily on dead hardwood trees. This fungus is a widely cultivated and important edible mushroom in its native range of eastern Asia, which includes eastern Russia, China, Taiwan, Korea, and Japan (Miyazawa et al., 2011). GOM was intentionally imported into North America in the1990s and continues to be sold in popular mushroom growing kits due to its easy cultivation, rapid growth, large yields, vibrant yellow caps, and distinctive nutty flavor. Around 2010, GOM escaped into the forests of North America and started spreading, rapidly expanding its range. Since then, GOM has been found in at least 25 American States and 2 Canadian provinces: Ontario and Quebec. Under a warming climate, models predict that this fungus will continue to expand its range from its current distribution in middle and northeastern North America (Veerabahu et al., 2025). GOM is a strong competitor and can reduce native fungal diversity in the trees it colonizes by 50% on average, modifying community composition and potentially affecting ecosystem decomposition rates (Veerabahu et al., 2025)
The focus on this description is to identify the fruiting body, or mushroom, of the Golden Oyster Mushroom, which is the most visible feature. Most of the year the fungus exists as a mycelium and is virtually undetectable.
Identifying Golden Oyster Mushroom
Cap: GOM can be identified by its cap which is bright yellow and vibrant when young and fades to a pale yellow or white with age (Singer 1943). Caps are typically 2 to 8 centimeters in diameter and have a central depression which can give them a funnel-like shape when mature. When young, the edges are curved but can flare or become wavy as they age.
Stem and gills: The stems of GOM are white, subcentral, branched, and joined together at the base since these mushrooms tend to grow in large clusters (Bruce, 2018). As a result of cluster density, stems are usually curved or bent. An identifying feature of this fungus is its deterrent white gills; the gills attach directly to the mushroom stem and run down its length.
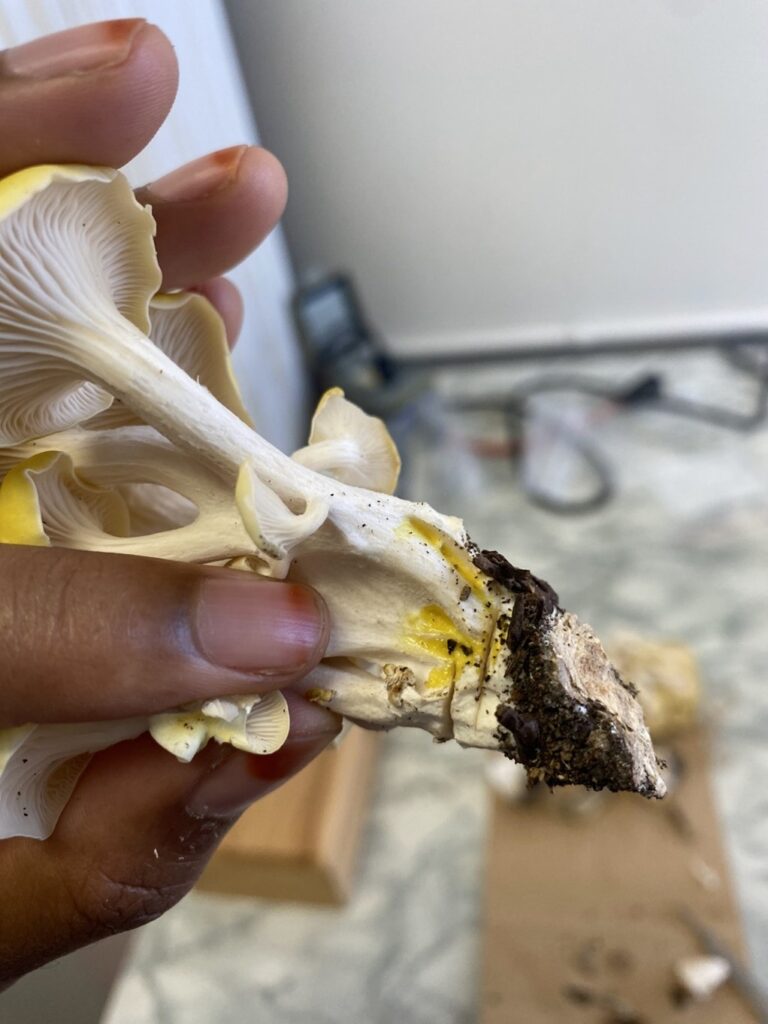
Spores: Known for a heavy spore load, gilled mushrooms, like GOM, can produce and release billions of spores a day (Hassett et al., 2015). GOM’s spores, like the spores of other mushroom species, are microscopic and invisible to the naked eye. Under a microscope GOM’s spores will appear whitish-pink, ellipsoid shaped, and 5–10 micrometers in length.
Odour: Strong unpleasant odour when past maturity. When fresh, GOM has been described by foragers to have a mild pleasant odour ranging from a light earthy scent to a watermelon/fruity scent to a seafood or fishy aroma.

There are a few look-a-like fungal species that can be mistaken for GOM. One species is the toxic Jack-o’-Lantern mushroom (Omphalotus illudens). These mushrooms are typically darker in colour (more orange than yellow) and have gills that only run partway down the stem, unlike GOM who have decurrent gills. Jack-o’-Lanterns also grow in larger clusters and have the ability to glow in the dark.

Another common look-alike species are honey mushrooms (Armillaria mellea) which have a similar yellow-brown coloration and also grow in clumps. This species however has slightly scaly caps, and its gills are not strongly deterrent.

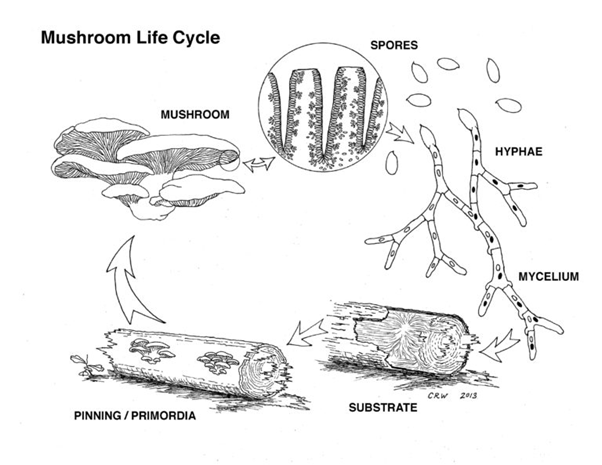
The life cycle of an oyster mushroom, like other basidiomycetes, starts with the release of basidiospores from the fungus’s mature fruiting body (mushroom) and the germination of these spores in a suitable substrate (typically decaying hardwood species like elm, oak, maple, or black cherry). If the environmental conditions are favorable, the basidiospores will germinate to form hyphae, thread-like structures that are the building blocks of fungal growth. When compatible hyphae meet, they fuse and over time form a complex network known as a mycelium (Nwankwegu et al., 2025). GOM will spend the majority of its existence as a mycelium network, hidden beneath soil or wood and invisible to the naked eye, and the visible reproductive organ, the mushroom, will only be visible for a few weeks to allow the production and release of spores. The mycelium network will continue to grow and expand, anchoring the fungus in its substrate and helping it absorb nutrients from the environment. When environmental conditions, including temperature, humidity, and oxygen levels are optimal, the mycelium will begin to develop its fruiting body (Martínez-Carrera, 1998; Nwankwegu et al., 2025). First through the production of hyphal knots and then primordia, a mature mushroom composed of a cap, stem, and gills finally develops. The basidia under the gills of this mature fruiting body are ready to release millions of whitish coloured basidiospores to restart the fungal life cycle.
Golden oyster mushrooms are wood decay fungi and are commonly found growing on dead deciduous hardwood species including oak, elm, and beech but have also been spotted on black cherry, maple, ash, cottonwoods, mulch, and straw (Veerabahu et al., 2025). They have a strong preference for dead elm trees and thrive in warm, humid deciduous hardwood forests. The distinct yellow fruiting body of the mushroom can be seen from May to September.
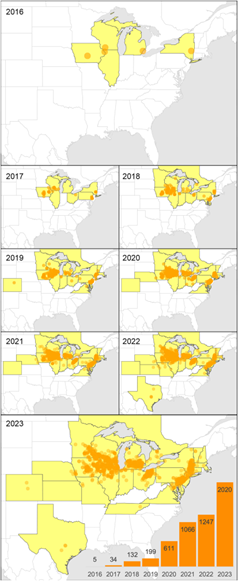
The native range of GOM is eastern Asian, including China, Taiwan, Japan, and eastern Russia. Thought to have escaped into the North American forests in 2010 following their intentional importation as cultivated edible mushrooms in the United States in the early 2000s, the first wild specimens were documented as single observations in 2013 (Athens, NY), 2014 (Madison, WI) and 2015 (OH) (Veerabahu et al., 2025). Since these first reports, 12,000 reports of GOM have been logged on iNaturalist in North America and the fungus has been documented in 25 middle and northeastern American states, and two Canadian provinces (ON and QC) (Hibbett et al., 2026; Veerabahu et al., 2025). Researchers have estimated that the species range now spans approximately 2 million km2. Due to their primary association with American elm trees, GOM’s current distribution in North America is similar to the distribution of this tree species. A map of GOM’s rapid spread based on community science observations submitted through iNaturalist and MushroomObserver platforms is available below. According to climate model predictions, GOM’s range will continue to expand under a warming climate to include many other areas in North America, such as Pacific hardwood forests, Alaska, Appalachia, and parts of Mexico (Veerabahu et al., 2025).
Ecological
GOM isn’t known to be a risk to living trees but does have an important impact on the species diversity and community composition of native wood-inhabiting fungal species. A recent study found that elm trees colonized by GOM hosted about half of the fungal species of trees without GOM, with GOM host trees harbouring 22 fungal species and non-hosts harbouring 40 fungal species (Veerabahu et al., 2025). GOM colonization not only affects the diversity of the fungal community on trees, but also its composition, lowering species richness and increasing competition with native fungi. This competition with other decomposers could have profound impacts on entire forest ecosystems. GOM belongs to a group of fungi called white rot fungi which, due to their efficient ability to decompose lignin, a key structural component of wood and bark, release more carbon dioxide into the atmosphere than brown rot fungi, another group which cannot digest this component (Simpson et al., 2024). A key preoccupation is that GOM is modifying the community composition of wood-dwelling fungi in North American hardwood forests and may be shifting the balance towards increasing presence of white rot fungi. As a member of the white rot fungus group, GOM’s range expansion and aggressive competition with native fungal species may be associated with increased rates of wood decay and CO2 emission, leading to unpredictable and cascading ecosystem effects including loss of habitat for insects, birds, and small mammals.
Economic
Although GOM doesn’t pose a risk to living trees and thus has minimal impacts on the forestry sector, its rapid spread and competitive features pose a threat to native wood dwelling fungi. Loss of important native fungal species will decrease the genetic variability needed for fungi and forests to adapt to changing climates and perform essential ecosystem functions, such as carbon sequestration and nutrient cycling, services that are valued in the trillions globally (Costanza et al.,1997). The potential discovery of medicinal compounds produced by fungi will also be lost with a decline in native fungal diversity.
Social
Competition between the invasive GOM and native fungal species may threaten edible native species, changing the landscape and available mushrooms for foragers. Conversely, for novice foragers, GOM represents an accessible entry point into foraging since it is easy to find and correctly identify, helping teach and document the threat of invasive mushrooms.
Management
There are currently no management strategies available to limit the spread of GOM but there are some suggestions in place for foragers, gardeners, and hobbyists. The most direct and effective way to minimize the spread would be to stop the sale of golden oyster mushroom growing kits and exclusively sell native or non-invasive mushroom kits. Gardeners and hobbyists are encouraged to purchase and grow native, locally sourced mushrooms. If GOM kits are purchased, grow kits must be disposed of responsibly. Used kits should not be composted or dumped outside since they may still contain GOM mycelium which can rapidly spread into the natural environment. Instead, kits should be sealed in plastic bags, left in the sun to sterilize the remaining traces of GOM, and placed in the garbage. If spotted in the natural environment, citizens are encouraged to report their findings on platforms such as iNaturalist, EDDMapS, or Mushroom Observer to help researchers track the spread. In the case of species identification, a useful resource for reporting a sighting is EDDMapS (https://www.eddmaps.org/). This user-friendly resource allows community members to report sightings of invasive species that are then confirmed by trained taxonomists who will identify the species via pictures from community members. This resource is accessible to anyone who wants to know what invasive species may be present in their area. Although not a large-scale solution, since these are edible mushrooms, foragers are encouraged to harvest them but only after carefully confirming their identity.
Articles and Research
Bruce, A.L. (2018). Population genomic insights into the establishment of non-native golden oyster mushrooms (Pleurotus citrinopileatus) in the United States [Master’s thesis, University of Wisconsin-La Crosse]. UW-L Theses & Dissertations.
Costanza, R., d’Arge, R., de Groot, R. et al. The value of the world’s ecosystem services and natural capital. Nature 387, 253–260 (1997)
Hassett, M. O., Fischer, M. W., & Money, N. P. (2015). Mushrooms as Rainmakers: How Spores Act as Nuclei for Raindrops. PloS one, 10(10), e0140407.
Hibbett, D. S., Prabhu, P., & Tabima, J. F. (2026). Invasion biology: The golden oyster mushroom in North America. Current biology : CB, 36(1), R12–R14.
Lee, C. H., Lee, Y. Y., Chang, Y. C., Pon, W. L., Lee, S. P., Wali, N., Nakazawa, T., Honda, Y., Shie, J. J., & Hsueh, Y. P. (2023). A carnivorous mushroom paralyzes and kills nematodes via a volatile ketone. Science advances, 9(3)
Martínez-Carrera, D. 1998. Oyster mushrooms. McGraw-Hill Yearbook of Science & Technology 1999. Ed.: M. D. Licker. McGraw-Hill, Inc., New York. Pp. 242-245. ISBN 0-07-052625-7 (447 pp.)
Nwankwegu, A. S., Hongsanan, S., Nwankwegu, U. P., & Xie, N. (2025). Exploring the Critical Environmental Optima and Biotechnological Prospects of Fungal Fruiting Bodies. Microbial biotechnology, 18(8), e70210.
Miyazawa, M., Dejima, Y., Takahashi, T., Matsuda, N., & Ishikawa, R. (2011). Characteristic Odor Components of Essential Oil from Dried Fruiting Bodies of Golden Oyster Mushroom (Pleurotus citrinopileatus). Journal of Essential Oil Research, 23(3), 58–63.
Simpson, H.J., Andrew, C., Skrede, I., Kauserud, H. and Schilling, J.S. (2024), Global field collection data confirm an affinity of brown rot fungi for coniferous habitats and substrates. New Phytol, 242: 2775-2786.
Singer, R. 1943: Das System der Agaricales. III. Annales Mycologici 41(1-3): 1-189
Veerabahu, A., Banik, M. T., Lindner, D. L., Pringle, A., & Jusino, M. A. (2025). Invasive golden oyster mushrooms are disrupting native fungal communities as they spread throughout North America. Current biology : CB, 35(16), 3994–4002.e4
